Division of inferior pulmonary ligament did not impact on the postoperative recovery or recurrence in patients undergoing video-assisted thoracoscopic surgery for primary spontaneous pneumothorax
Introduction
Primary spontaneous pneumothorax (PSP) has been a worldwide health problem, with the incidence around 18–28/100,000 cases per annum for men and 1.2–6/100,000 for women (1). Video-assisted thoracoscopic surgery (VATS) has been proved as a safe and reproducible approach for PSP. The recurrence rate of PSP was reduced to 10–20% with bullectomy alone, and 1–6% with bullectomy and chemical or mechanical pleurodesis (1-5). The occurrence of PSP mostly results from rupture of apical blebs or bulla. Successful treatment of PSP lies in elimination of air-leaking blebs or bulla, and ablation of apical pleural space. Besides blebectomy, bullectomy, and pleurodesis, division of inferior pulmonary ligament (IPL), which has been reported to prevent postoperative complications associated with residual pleural space and prolonged air leak, might contribute to the reduction of postoperative air leakage and recurrence in patients undergoing VATS for PSP (6). Herein we retrospectively analyzed the impact of IPL division on the surgical outcome of VATS for PSP.
Methods
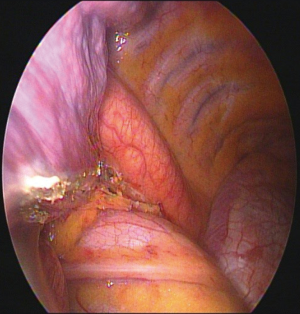
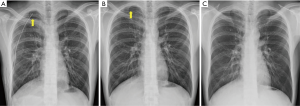
Patients younger than 30 undergoing VATS for PSP between Dec. in 2013 and Sep. in 2015 were enrolled in this study. All these patients underwent VATS blebectomy or bullectomy followed by mechanical and chemical pleurodesis with tetracycline 1000mg. Patients with odd chart numbers underwent additional procedure as division of IPL, and were classified as rIPL group demonstrated in Figure 1. Patients who did not undergo division of IPL were classified as control group. A 20 Fr chest tube was placed in the pleural space postoperatively, and negative pressure suction with −10 cmH2O was applied only if there was air leakage. The chest tube was removed when there was no more air leakage. The 1st postoperative chest plain film (CXR) was done on the 1st postoperative day (POD1), the 2nd was done on POD 7, and the 3rd was done at the clinic 2 months after the surgery (Figure 2). Recurrence was defined as postoperative ipsilateral pneumothorax, whereas contralateral pneumothorax was defined as new episode of pneumothorax. The follow-up period ranged from 44 to 67 months. Surgical outcomes such as operative time, days of chest tube drainage, postoperative recurrence, and the residual pleural space on the postoperative serial CXRs were included for analysis.
Statistic
Categorical data were presented as number (percentages), and non-categorical data as the mean ± standard deviation. Comparison of categorical variables between the study groups was performed using Chi square test, whereas comparison of continuous variables was performed using Student’s t-test. The Kaplan-Meier method was used to calculate recurrence-free survival (RFS). A P value of ≤0.05 was set to indicate statistical significance. SPSS system (IBM SPSS Statistics, Version 22.0, Armonk, NY, USA) was used for statistical analysis.
Results
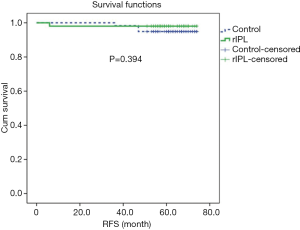
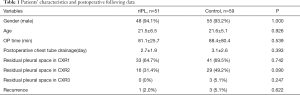
A total of 110 patients underwent VATS wedge resection and pleurodesis for PSP between Oct. 2013 and Dec. 2015. Fifty-one of the 110 patients underwent division of IPL as the rIPL group, and the rest 59 did not (control group). Patients in both groups were male predominant [rIPL 48 (94.1%) versus control 55 (93.2%), P=1.000] and at young age (rIPL 21.5±6.5 years versus control 21.6±5.1 year, P=0.926). Patient demographics and postoperative follow-up information were listed in Table 1. The operative time was longer in the rIPL group, and the difference was not significant (rIPL 81.1±25.7 min versus control 88.4±80.4 min, P=0.539). The period of time for chest tube drainage was not different between the two groups (rIPL 2.7±1.9 days versus control 3.1±2.6 days, P=0.393). The patient number with visible residual pleural space on POD1 CXR in each group was 33 (64.7%) in the rIPL group and 41 (69.5%) in the control group (P=0.742). The patient number with visible residual pleural space on POD7 CXR in each group was 16 (31.4%) in the rIPL group and 29 (49.2%) in the control group (P=0.090). There were 3 patients (5.1%) with visible residual pleural space in the control group and none in the rIPL group on the 2-month follow-up CXR (P=0.247). There was no postoperative hemothorax in each group or complication necessitating invasive procedure. One patient (2.0%) in the rIPL group and 3 (5.1%) in the control group had recurrence. The Kaplan-Meier curve for recurrence-free period revealed no difference (P=0.394) (Figure 3).
Full table
Discussion
Our surgical results demonstrated that division of IPL did not influence the postoperative recovery or recurrence in patients undergoing VATS for PSP. The CXR on POD1 revealed residual pleural space in more than 60% in each group. The residual pleural space remained visible on the POD7 CXR, and mostly did not impact on the decision of postoperative care and chest drain management. The residual pleural space was compensated eventually because of hyperinflation of the remaining parenchyma.
The IPL is a thin structure of fused triangular-shaped sheet of parietal and visceral pleura, connecting the inferior pulmonary vein and the dome of hemidiaphragm. After lung resection, the pleural space for remaining lung increases, and then gradually diminished because of the compensation mechanism consisting of mediastinal shifting, diaphragm elevation, and overexpansion of the remaining lung parenchyma. It has been reported that division of IPL increased operative time but had no impact on the postoperative pulmonary expansion and recurrence rate, which is similar with our results (7). Thoracic surgeons supporting division of IPL after upper lobectomy suggested that this maneuver increased the mobility of the remaining lung parenchyma and decreased the likelihood of postoperative air leakage (6). On the contrary, division of IPL after upper lobectomy might change the angle of bronchus and lead to bronchial distortion, bronchial stenosis, or atelectasis (8-10). Moreover, preservation of IPL might preserve the postoperative pulmonary function (10). These aforementioned results concluded that division of IPL did not pose substantial advantage on the postoperative recovery (9-12).
In our series, the postoperative hospital stay for PSP was 2 to 3 days, and the postoperative recurrence rate was low compared to the literature report. The potential benefit of IPL division might not be clinically evident in patients undergoing wedge resection with pleurodesis for PSP. The extent of surgery for PSP was limited without upper lobectomy, and the remaining upper lobe parenchyma prevented bronchial distortion and the occurrence of a large residual pleural space. Division of IPL might therefore contribute little to the postoperative recovery of VATS wedge resection for PSP, and the procedure per se might injury the inferior pulmonary vein or parenchyma of lower lobe.
Conclusions
VATS IPL division for PSP does not impact on the postoperative recovery or reduction of air leak and recurrence.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Jin-Shing Chen, Ke-Cheng Chen and Mong-Wei Lin) for the series “VATS: Primary Spontaneous Pneumothorax” published in Video-Assisted Thoracic Surgery. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/vats.2019.08.03). The series “VATS: Primary Spontaneous Pneumothorax” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by Institutional Review Board of National Cheng Kung University Hospital (B-ER-104-141) and written informed consent was obtained from all patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- MacDuff A, Arnold A, Harvey J. Management of spontaneous pneumothorax: British Thoracic Society Pleural Disease Guideline 2010. Thorax 2010;65:ii18-31. [Crossref] [PubMed]
- Mithiran H, Leow L, Ong K, et al. Video-Assisted Thoracic Surgery (VATS) Talc Pleurodesis Versus Pleurectomy for Primary Spontaneous Pneumothorax: A Large Single-Centre Study with No Conversion. World J Surg 2019;43:2099-2105. [Crossref] [PubMed]
- Ng C, Maier HT, Kocher F, et al. VATS Partial Pleurectomy Versus VATS Pleural Abrasion: Significant Reduction in Pneumothorax Recurrence Rates After Pleurectomy. World J Surg 2018;42:3256-62. [Crossref] [PubMed]
- Olesen WH, Katballe N, Sindby JE, et al. Surgical treatment versus conventional chest tube drainage in primary spontaneous pneumothorax: a randomized controlled trial. Eur J Cardiothorac Surg 2018;54:113-21. [Crossref] [PubMed]
- Sepehripour AH, Nasir A, Shah R. Does mechanical pleurodesis result in better outcomes than chemical pleurodesis for recurrent primary spontaneous pneumothorax? Interact Cardiovasc Thorac Surg 2012;14:307-11. [Crossref] [PubMed]
- Usuda K, Sagawa M, Aikawa H, et al. Do Japanese thoracic surgeons think that dissection of the pulmonary ligament is necessary after an upper lobectomy? Surg Today 2010;40:1097-9. [Crossref] [PubMed]
- Yan Joyce Chan W, Yu P, Lau R, et al. Inferior Pulmonary Ligament Release During Surgery for Primary Pneumothorax: Time-consuming Without Reduced Recurrence. Heart, Lung and Circulation 2018;27:S552. [Crossref]
- Bu L, Yang AR, Peng H, et al. Dividing inferior pulmonary ligament may change the bronchial angle. J Surg Res 2016;201:208-12. [Crossref] [PubMed]
- Kim DH, Moon DH, Kim HR, et al. Effect of inferior pulmonary ligament division on residual lung volume and function after a right upper lobectomy. Interact Cardiovasc Thorac Surg 2019; [Epub ahead of print]. [Crossref] [PubMed]
- Seok Y, Yi E, Cho S, et al. Perioperative outcomes of upper lobectomy according to preservation or division of the inferior pulmonary ligament. J Thorac Dis 2015;7:2033-40. [PubMed]
- Khanbhai M, Dunning J, Yap KH, et al. Dissection of the pulmonary ligament during upper lobectomy: is it necessary? Interact Cardiovasc Thorac Surg 2013;17:403-6. [Crossref] [PubMed]
- Matsuoka H, Nakamura H, Nishio W, et al. Division of the pulmonary ligament after upper lobectomy is less effective for the obliteration of dead space than leaving it intact. Surg Today 2004;34:498-500. [Crossref] [PubMed]
Cite this article as: Chang CC, Lai WW, Tseng YL, Yen YT. Division of inferior pulmonary ligament did not impact on the postoperative recovery or recurrence in patients undergoing video-assisted thoracoscopic surgery for primary spontaneous pneumothorax. Video-assist Thorac Surg 2019;4:20.