
Is routine dissection of lymph node station 4L clinically significant in left lung NSCLC?
I read with special interest the article entitled “Clinical significance of 4L lymph node dissection in left lung cancer” published in Journal of Clinical Oncology in October 2018 (1). The authors did a retrospective analysis of their patients who underwent lung parenchyma resection for non-small cell lung cancer (NSCLC) of the left lung during the 5-year period 2005–2009. The main conclusions of their study were that dissection of the 4L lymph node station revealed a quite high rate of involvement by the tumor (20.9%), while 4L lymph node dissection was found to be an independent prognostic factor for improved disease-free survival (DFS) and overall survival (OS) in left lung NSCLC. In case the above-mentioned results will be reproduced in future studies, dissection of the 4L lymph node station will be definitely considered an essential step in the surgical management of left lung NSCLC which will affect the clinical practice and the guidelines for lymph node dissection of left lung NSCLC.
It is questionable why only a small percentage (21.1%) of the 657 patients who entered the analysis underwent dissection of the 4L nodal station. The main drawback of the study is the lack of criteria to proceed with 4L lymph node dissection in the 657 patients of the study. Propensity score matching can correct selection biases for a set of observed variables, indeed biases may remain after matching due to latent variables. In the article to be commented the source of possible bias are the criteria to proceed or not with dissection of 4L lymph node station in the study population. One’s could suggest that dissection of 4L nodes was possibly based on preoperative imaging suspicious for involvement of the 4L nodes or on the choice/experience of the responsible surgeon or on other clinical characteristics which are not included in the set of variables used for propensity matching. The tools used for preoperative N staging are not reported because probably all these data could not be available in a retrospective study including patients who underwent surgery more than 10 years ago.
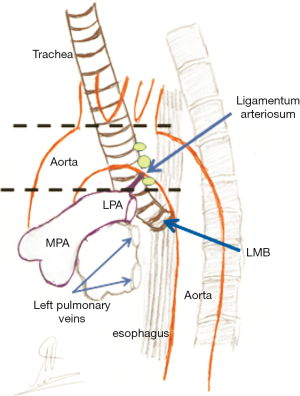
According to the currently in use nodal chart introduced by the International Association for the Study of Lung Cancer (IASLC), the 4L lymph node station includes nodes to the left of the left lateral border of the trachea and medial to the ligamentum arteriosum (Bottalo’s ligament, ductus arteriosus) (2). The upper border of the station is the upper margin of the aortic arch and the lower border is the upper rim of the left main pulmonary artery (2). According to the above nodal station definitions made by IASLC, the nodes of station 4L lie within the concavity of the aortic arch and behind the aortic arch as seen in Figure 1. Furthermore, it is well known from the past that 4L nodes are a less compact group and the lymph nodes can be found either, in close relation to the left recurrent laryngeal nerve or more anteriorly, forming a continuous link with the subaortic nodes in the region of ligamentum arteriosum (3,4).
Lymph drain of the left lung is also quite complicated and may follow different patterns according to the bronchopulmonary segment where the tumor is located (3-6). Left upper lobe tumors tend to metastasize to the aortopulmonary zone including subaortic (station 5) and para-aortic nodes (station 6) (3-5). Tumors which grow in the apicoposterior and anterior segments can also give metastases to the ipsilateral tracheobronchial nodes (station 4L) and to subcarinal nodes (station 7), while drainage to the contralateral tracheobronchial nodes (station 4R) in observed in 28% of the cases (3-5). Lingular growths have a similar pattern of lymph drainage, while infiltration of the contralateral nodes is observed in 25% of the cases (3,4). Tumors located in the superior segment of the left lower lobe metastasize to ipsilateral and contralateral mediastinal nodes; the rate of malignant spread to the contralateral nodes is surprisingly high (42%). The basal segments of the left lower lobe metastasize also to the contralateral nodes through initial involvement of the subcarinal nodes (3,4).
Lymph node station 4L is not easy to access because the nodes are well covered by the aortic arch. In addition, the nodes are found in close proximity with the recurrent laryngeal nerve which can be damaged during dissection of the nodes (3,4). The lymph nodes of the station 4L (named in the past the left superior tracheobronchial nodes) are located on the medial side of the subaortic nodes and they can be exposed and dissected after careful division of the ligamentum arteriosum. To gain access to the 4L nodes since the division of ligamentum arteriosum it is necessary to smoothly retract upwards the aortic arch and isthmus (3,7). Toker and coworkers reported in 2011 an alternative to the classical technique to access the 4L lymph node station through left thoracotomy. The authors propose to start with complete dissection of the station 5 nodes and then proceed with blunt dissection below the aortic arch by creating a tunnel between the arch, the left main bronchus, the left main pulmonary artery and the ligamentum arteriosus (8). Dissection of the 4L station by any technique or surgical access (open or thoracoscopic) is not an easy surgical maneuver and it should always be kept in mind that a permanent damage to the left recurrent nerve can be made (1,3,4). Surgical manipulations and retraction of a dilated or atherosclerotic aortic arch has the potential risk of aortic dissection or hematoma or of the embolization of atheromatic debris from the aortic wall. Injury to the thoracic duct is another possible complication when dissecting the 4L lymph node station which can result in postoperative chyle fistula (1,3). In a recent article by Kostoulas and Papagiannopoulos, the authors state that dissection of station 4L is not routinely performed during left-sided pulmonary resections due to the limited exposure of the station which is covered by the aortic arch (9). On the same wavelength, Wright does not include station 4L in the plan of lymph node dissection during left-sided VATS resections (10). In my experience, removal of the 4L nodes is easier to accomplish in case of pneumonectomy because the main pulmonary artery is centrally ligated, liberating that way some extra space in the aortopulmonary window. In addition, the access is further facilitated by retracting downwards the left main bronchus since the division of pulmonary vessels and freeing the bronchus from the peribronchial tissues.
The available clinical guidelines for the surgical treatment of NSCLC recommend to proceed with complete lymph node dissection or systematic sampling including at least 3 mediastinal lymph node stations in order to define with accuracy the pN stage of the tumor (11-13). The guidelines published by the British Thoracic Society in 2010 recommends that “at least six lymph node stations should be removed or sampled before the confirmation of pN0 status”; three out of the six removed nodal stations should be mediastinal, including always the subcarinal nodes (station 7) (11). The European Society of Thoracic Surgeons (ESTS) propose resection or sampling of lymph node stations No 5, 6 and 4L as the minimal requirement for all tumors located in the left lung (7). In a review published by Whitson and coworkers in 2007, the authors propose to examine at least 10 mediastinal nodes obtained from three or more mediastinal lymph node stations (14). The important questions that arise are:
- Which should be the three mediastinal stations to be sampled according to the lobe where the tumor is located? No consistency exists in the literature.
- Which should be the stations to be sampled for tumors located in the left main bronchus or in the secondary carina or for those which cross the fissure involving both lobes? It is quite simple to suggest lobar specific dissection or sampling (i.e., stations 5, 6 and 7 for left upper lobe tumors and stations 7, 8, 9 for left lower lobe tumors) (9), however, should we ignore station 4L because it is not easy to access?
In a previous study published by Wang YX and coworkers in 2015, the infiltration rate of the 4L nodes from tumors of the left upper or left lower lobe was found to be very low (2.3% and 1% respectively) and consequently we can exclude station 4L from the standard plan of mediastinal lymph node dissection in left lung NSCLC, unless specific indications exist (6,9). In the article that is commented in this editorial, invasion of the station 4L in left lung NSCLC was found to be associated with concurrent metastases to other mediastinal lymph node stations, mainly stations 5, 6 and 7. Patients with metastases to multiple lymph node stations are not the ideal surgical candidates to draw conclusions for the importance on OS and DFS of the routine dissection of a single nodal station. Possible involvement of the station 4L in left lung NSCLC has to be ruled out before surgery by using PET/CT scan, EBUS and Video-Mediastinoscopy according to the guidelines and the simple algorithm for mediastinal staging proposed by the ESTS and the European Society of Medical Oncology (13,15). Complete clearance of the 4L nodes could be reserved only for surgical procedures performed for the radical resection of downstaged left lung tumors, where suspicious or proven involvement of the station 4L was detected before induction treatment.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Video-Assisted Thoracic Surgery. The article did not undergo external peer review.
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/vats.2019.01.01). The author has no conflicts of interest to declare.
Ethical Statement: The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Wang YN, Yao S, Wang CL, et al. Clinical significance of 4L lymph node dissection in left lung cancer. J Clin Oncol 2018;36:2935-42. [Crossref] [PubMed]
- Rusch VW, Asamura H, Watanabe H, et al. The IASLC lung cancer staging project: a proposal for a new international lymph node map in the forthcoming seventh edition of the TNM classification for lung cancer. J Thorac Oncol 2009;4:568-77.
- Nohl-Oser HC. Lymphatics of the lung. In: Nohl-Oser HC, Nissen R, Schreiber HW (eds). Surgery of the lung. Georg Thieme Verlag, Stuttgart-New York, 1980:49-53.
- Shields TW. Lymphatic drainage of the lungs to the mediastinal lymph nodes. In: Shields TW, LoCicero J, Ponn RB (eds). General Thoracic Surgery, 5th Edition. Lippincott Williams & Wilkins, Philadelphia, 2000: 87-90.
- Kotoulas CS, Foroulis CN, Kostikas K, et al. Involvement of lymphatic metastatic spread in non-small cell lung cancer accordingly to the primary cancer location. Lung Cancer 2004;44:183-91. [Crossref] [PubMed]
- Wang YX, Li BS, Huang W, et al. Pattern of lymph node metastases and its implication in radiotherapeutic clinical target volume in patients with non-small-cell lung cancer: a study of 2062 cases. Br J Radiol 2015;88:20140288 [Crossref] [PubMed]
- Lardinois D, De Leyn P, Van Schil P, et al. ESTS guidelines for intraoperative lymph node staging in non-small cell lung cancer. Eur J Cardiothorac Surg 2006;30:787-92. [Crossref] [PubMed]
- Toker A, Tanju S, Ziyade S, et al. Alternative paratracheal lymph node dissection in left-sided hilar lung cancer patients: comparing the number of lymph nodes dissected to the number of lymph nodes dissected in right-sided mediastinal dissections. Eur J Cardiothorac Surg 2011;39:974-80. [Crossref] [PubMed]
- Kostoulas N, Papagiannopoulos K. Mediastinal lymph node dissection in open thoracic surgery. Shanghai Chest 2017;1:22. [Crossref]
- Wright GM. VATS lymph node dissection. Ann Cardiothorac Surg 2012;1:102-3. [PubMed]
- Lim E, Baldwin D, Beckles M, et al. Guidelines on the radical management of patients with lung cancer. Thorax 2010;65:iii1-27. [Crossref] [PubMed]
- Detterbeck FC, Lewis SZ, Diekemper R, et al. Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:7S-37S.
- Postmus PE, Kerr KM, Oudkerk M, et al. Early and locally advanced non-small-cell lung cancer (NSCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2017;28:iv1-21. [Crossref] [PubMed]
- Whitson BA, Groth SS, Maddaus MA. Surgical assessment and intraoperative management of mediastinal lymph nodes in non-small cell lung cancer. Ann Thorac Surg 2007;84:1059-65. [Crossref] [PubMed]
- De Leyn P, Dooms C, Kuzdal J, et al. Revised ESTS guidelines for preoperative mediastinal lymph node staging for non-small-cell lung cancer. Eur J Cardiothorac Surg 2014;45:787-98. [Crossref] [PubMed]
Cite this article as: Foroulis CN. Is routine dissection of lymph node station 4L clinically significant in left lung NSCLC? Video-assist Thorac Surg 2019;4:2.


