
Nonintubated uniportal nonresectional videothoracoscopic lung volume reduction surgery
Introduction
Lung volume reduction surgery (LVRS) was originally theorized and performed as the surgical resection of the most severely emphysematous target area (1,2). In properly selected patients, this technique proved durable improvement in exercise capacity, respiratory function, overall survival, quality of life and nutritional status (3,4). We originally performed this procedure under multi-portal video-assisted thoracic surgery (VATS) in general anesthesia and single lung ventilation (4-6).
In 2001, one of us (Tommaso Claudio Mineo) introduced a novel personal technique of LVRS entailing the plication of the most emphysematous lung regions. The operation was directed at reducing 20–30% of the lung volume by plicating functionally useless and hyperinflated lung tissue (7). Functional outcomes resulted as valid as the resectional LVRS, whereas nonintubated nonresectional procedure was associated with less morbidity and more rapid recovery mainly due to the avoidance of general anesthesia-related adverse effects (8,9). Initially, the procedure was conducted under thoracic epidural anesthesia (TEA) and multi-portal VATS (7,10). More recently, we have performed the same technique through an uniportal approach under intercostal block.
Hereby, we critically analyzed our entire experience of nonintubated nonresectional LVRS after a very long period of follow up and we compare the outcomes of the uniportal group and the multi-portal group.
Patients and methods
After Institutional Review Board approval and informed consent 108 consecutive patients with moderate-to-severe emphysema who underwent nonresectional LVRS under nonintubated anesthesia between January 2001 and October 2015, were retrospectively studied. In 77 instances, the procedure was accomplished through a conventional multi-portal VATS approach. The last 31 patients were approached using a uniportal technique.
Preoperative workup
Main tests for surgical inclusion are shown in Table 1. Radiologic study included digital inspiratory and expiratory chest radiographs performed in posteroanterior and lateral views and high resolution computed tomography of the chest. Arterial blood gases were analyzed at rest, on room air. Pulmonary function tests were performed after administration of 2 puffs of aerosolized salbutamol. Static lung volumes were determined by plethysmography and diffusing capacity for carbon monoxide was assessed by the single-breath technique. Exercise tolerance was assessed by standard 6-min walking test and by maximal incremental treadmill test. The 6-min walking test was carried out with standardized encouragement and an oxygen supply titrated to maintain oxygen saturation above 90%. At baseline and during the follow-up visits dyspnea was rated according to the modified Medical Research Council Score (11). Quality-of-life was assessed with the Medical Outcomes Study Short Form (SF)-36 item (12) and St. George’s Respiratory Questionnaire General Score (best =0, worst =100) (13).
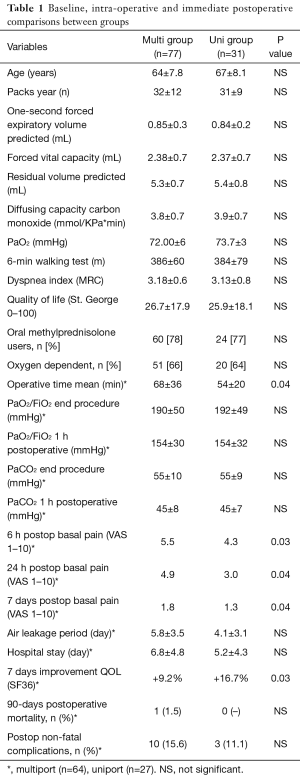
Full table
Main inclusion criteria were those applied for standard LVRS (1-3). They included severe disability despite maximized medical care (severe dyspnea, short distance at 6 min walking test, poor quality of life score), massive hyperinflation (residual volume more than 180% predicted) with upper lobes prevailing target areas and asymmetric emphysema at radiological examination, marked bronchial obstruction despite bronchodilator (forced expiratory volume in 1 second less than 40% predicted). In addition, patients should demonstrate smoking cessation and capability of performing a respiratory rehabilitation program. Initially, age more than 75 years and absence of severe comorbidity were strict exclusion criteria. However, with the increment of the experience and the shift to uniportal procedure these last prerequisites became less strict and a progressive higher number of elderly and/or comorbidity patients were considered for LVRS.
Furthermore, all patients must fit the general criteria for VATS operations: anamnestic or imaging absence of pleural adhesion, normal coagulation tests and absence of other bleeding disorders, non-obese patients (mass body index <30 kg/m2).
Finally, supplementary requisites deal with the feasibility of the operation under non-intubated anesthesia. These rules entail the easy accessibility to the airways, no risk of intraoperative seizures as well as stable and cooperative psychological profile. This status was always preoperatively assessed by a specialist with dedicated tests and with an interview. Every patient scheduled for tubeless surgery is now processed with new developed questionnaires such as Profile of Mood States and Mini Mental State Examination (14), which require a 30-min time. Intraoperative evaluation of the tolerance and state of consciousness was done with the Richmond Agitation-Sedation Scale (15). Preliminary data of the study are now available and we found that a combination of provides a better prediction of the tubeless procedure tolerance. No panic has been found in patients with low combined scores in these two questionnaires.
Indications to LVRS always emerged after case-by-case discussion within a panel of specialists including surgeons, anesthesiologist, pulmonologist, intensive-care specialist, physiotherapist and psychologist.
Multi-portal technique
In patients undergoing multi-portal technique (n=77), the anesthesia was administrated through a thoracic epidural catheter inserted at T4 after premedication of 7.5 mg midazolam. In the operating room, patients received a continuous infusion of ropivacaine 0.5% and sufentanil 1.66 µg/mL into the epidural space. In some instance, a topical vagal blockade was also performed.
The operation is performed in lateral decubitus and four flexible thoracoscopic trocars were inserted, one for the operative thoracoscope usually placed in the sixth intercostal space along the midaxillary line and the others placed in the third and fifth intercostal space along anterior axillary lines and in the fourth intercostal space along the posterior axillary line for the no-knife devices and, when needed, a gauze pad mounted on a ring-forceps to contrast lung movements during breathing.
At the end of the procedure, 28 CH chest tube was inserted through the lowest incision. Lung re-expansion was achieved under thoracoscopic vision by asking the patient to breathe deeply and cough repeatedly. The epidural catheter was usually removed on postoperative day 1.
Uniportal technique
All patients undergo a local anesthesia, after insertion of venous and radial artery catheters. An aerosolized 5 mL solution of 2% lidocaine is administered for 5 min in order to avoid cough reflex. The intercostal block is accomplished by injection of 10 mL 7.5% ropivacaine around the nerve of the selected space after separate local infiltration of 10 mL 2% lidocaine, for achieving both rapid onset and long duration of the analgesic effect. Site of infiltration is done along the space selected for uniportal VATS and included subcutaneous layers, intercostal nerves and parietal pleura. In a few cases benzodiazepine (midazolam 0.03–0.1 mg/kg) or opioids (remifentanil 15 µg/kg/min) are intravenously supplemented during lung manipulation. The chest tube with a water seal system is always kept ready on the instrument table.
The operation is performed in lateral decubitus. The single port of 30–40 mm is carried out along the space judged the most suitable to reach the foreseen area. The intercostal space is enlarged by the self-standing cylinder device (Alexis®, Applied Medical, Rancho Santa Margarita, USA) that works both as port protector and muscle retractor. This unique port must permit the introduction of all required instruments. Generally, the port was placed in the fourth intercostal spaces because the upper lobe of the lung is the most affected area. This incision allows the introduction of the operative thoracoscope, the 45-mm “no-knife” endostapler with 3.5 mm cartridges and single ring forceps. Plication of target areas is performed by no-knife stapling devices rising folds of tissue and suturing them at the basis (Figure 1). After the plication in some instances we use to apply with the help of a nebulizer a liquid cyanoacrylate (Glubran II®) on lung tissue in order to minimize the risk of air leaks.
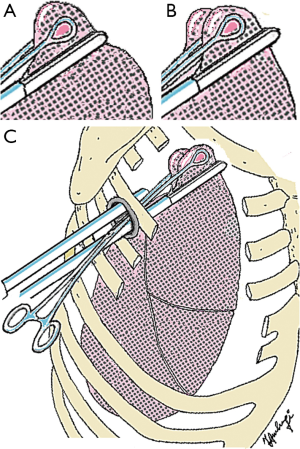
At the end of the procedure one 28 CH chest tube is collocated through the posterior end of the incision. Muscle sutures are tightened after asking the patient to breathe deeply or cough to achieve maximal lung re-expansion.
Postoperative care
All patients soon after the procedure are shifted to recovery where are evaluated by the same anesthesiologist in charge of them during the operation. Everybody undergo a chest X-ray documenting lack of significant pneumothorax or hemothorax. After a couple of hours, the patients are send to the Emphysema Unit in the ward where they are allowed to drink, to consume soft meals and to initial physiotherapy program. Patient are usually discharged when air leak stops or at least when it can be managed with the sole use of Heimlich valve.
Follow up
Early results were always re-discussed within the same panel group who set the indications. Respiratory and functional parameters were evaluated yearly. Patients undergoing unilateral treatment were considered for contralateral LVRS, when either spirometric values approximate to the baseline value or the patient became dissatisfied with the functional gain. In addition, in a cohort of bilaterally treated patients who eventually approximate to the disabled preoperative status who were not suitable candidates for lung transplantation and in whom high-resolution computed tomography showed severe emphysematous lung destruction that was prevailing in one upper lobe, redo LVRS was also considered.
Statistical evaluation
Descriptive statistics are presented as mean ± standard deviation (SD) while post-treatment changes were indicated as the mean percentage of the baseline value. Due to the relatively small sample size non-parametric tests for paired, and unpaired comparisons (Wilcoxon-sum rank and Mann-Whitney, respectively) were prudentially used (SPSS® 19.0 version, Chicago, IL, USA). Significance was set at P<0.05. Survivals were evaluated by Kaplan-Meier method (16), with the day of operation as starting point and the day of residual volume returned equal to baseline value or the day of death as endpoints. Significance test was assessed according to the Mantel log-rank test.
Results
The two groups (multi-portal and uniportal) were homogeneous for anagraphic and clinical data, whereas uniportal group showed a more elevate age 64 vs. 67 years (Table 2). Notably, 4 patients were more than 80 years old. We experienced a significantly shorter operative time for uniportal VATS (Table 2), this probably due to upgrade of the learning curve and shorter number of cuts. In uniportal group conversion rate to general anesthesia was 12.1% due to surgical reasons (n=2) and intolerance (n=2) and this was similar to that of multi-portal (11/77=14.3%).
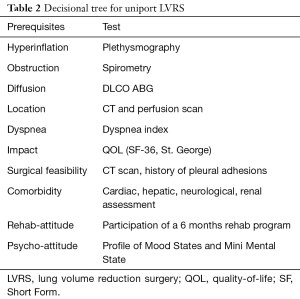
Full table
Early outcome
No significant difference was found between groups in oxygenation 1 h after surgery expressed by the ratio of arterial oxygen tension to fraction of inspired oxygen (PaO2/FiO2), and arterial carbon dioxide tension (PaCO2).
We experienced just one death within 90 days from multiport LVRS due to acute pneumonia and none in the uniportal group, yet no statistically different. Similarly, non-fatal complications rate was higher in multi (15.6%) than uniportal (11.1%) group.
Median hospital stay and median air leakage period resulted shorter in uniportal group even though not significantly (Table 1). Interestingly, we found a lesser median basal pain in the uniportal patients at both 6 h (4.3 vs. 5.5; P=0.03), 24 h (3.0 vs. 4.9; P=0.04) and 7 days (1.3 vs. 1.8; P=0.04) after surgery. This had an impact on 7 day-postoperative quality of life percentage improvement measured with physical component summary (+9.2% vs. 16.7%, P=0.03).
Long-term outcome
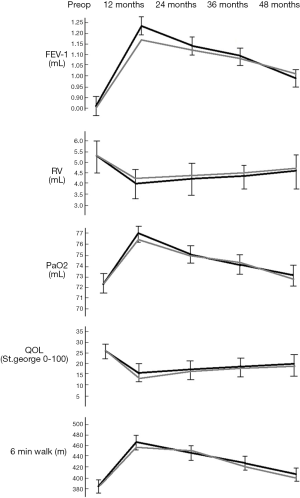
Mean follow up was 51±30 months for multi-portal and 42±24 months for the uniportal group, respectively. All respiratory and symptomatic parameters significantly improved in both groups with no intergroup significant difference (Figure 2). In 17 patients undergoing uniportal operation the LVRS was repeated on the other side after a mean period of 34±16 months. Residual volume persisted higher than preoperative values for more than 36 months in 24 patients. This evolution was not significantly different from that documented after the multi-portal procedure (Figure 2).
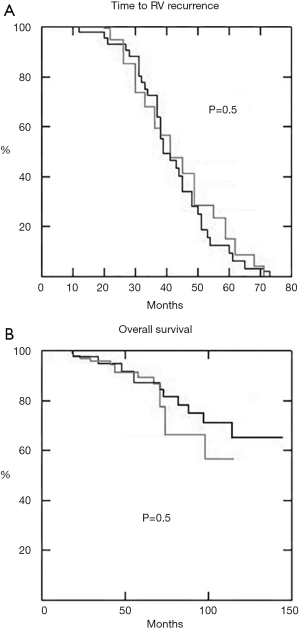
Analysis of long-term survival showed no statistical significant difference between uniportal and multi-portal groups in terms of both time-to-residual recurrence (Figure 3A) and overall survival (Figure 3B).
Comment
LVRS has becoming a reliable therapeutic opportunity in the treatment patients with upper-lobe predominant emphysema and impaired exercise capacity (17-20). Unfortunately, the irrefutable advantages derived from this operation are often concealed by the significant postoperative morbidity and mortality that remains quite elevate and hinders a wider diffusion of the procedure (21-22).
The introduction of nonintubated and nonresectional LVRS allowed a significant decrement of these postoperative complications. Initially multi-portal LVRS was safely and easily performed under TEA or local anesthesia (5,10). Now the possibility of performing this procedure through a single 2–4 cm long incision has consistently facilitated the acceptance of both the patients and the pulmonologists (9).
This study demonstrated a favorable improvement achieved by using the uniportal approach with a better tolerance for the patient and better immediate quality of life score. Notably, also lower 90-day postoperative mortality and non-fatal complications rate were better compared to multiport approach, yet not significant. The avoidance of epidural anesthesia can be considered a further advantage implying a broader amount of patients suitable for LVRS and less postoperative risk. Thus, the introduction of uniportal LVRS have progressively make possible to the fall of some the of the traditional threshold to LRVS with a simultaneous recruitment of individual previously deemed unfit for this kind of surgery. This renewed confidence implies that many patients are more rapidly proposed by physicians and pulmonologists to the surgeon and operated earlier than used to happen before.
No differences were found in long-term outcomes between uniportal and multi-portal groups and these results were not significantly different from those achieved with a traditional resectional procedure in general anesthesia. Indeed, even using the uniportal access and nonresectional technique, more than three quarters of the patients the residual volume persisted below the preoperative value for more than 36 months. Majority of the early recurrences of the residual volume were due to scant postoperative increment of the value despite the postoperative amelioration of the flows and the subjective symptomatic improvement. In another subset of patients, the rapid worsening of residual volume value was unpredictable and in this group we hypothesize a different genetic basis.
We acknowledge some limitation to this study. We think that major limitation of the study is represented by the retrospective non-randomized nature of the investigation, but this evident flaw can be counterbalanced by the consistent sample size collected in one single institution. At this regard, a prospective randomized double arm study is under the evaluation of our internal review board and ethics committee.
Our results allow us to consider that uniportal nonintubated nonresectional thoracoscopic LVRS is as safe and reliable as a multiport approach. Besides this operation is capable of significantly and durably ameliorating respiratory and symptomatic parameters with long-term result comparable to multi-portal procedure and to traditional resectional LVRS. Furthermore, the particular lower impact of this approach on general conditions will favor diffusion among elderly patients and patients with comorbidities, who have been traditionally ruled out from classic LVRS.
The true benefits and virtues of nonintubated uniVATS should be further verified in future prospective studies. Further investigations are necessary to clarify the applicability and benefits of this technique for specific patient groups.
Acknowledgments
Funding: This research was granted by the MURST COFIN grants No. 9906274194-06 and 2001061191-001, CNR CU0100935CT26 2002 and Centro di Eccellenza 2001.
Footnote
Provenance and peer review: The article was commissioned by the editorial office, Video-Assisted Thoracic Surgery for the series "Non-intubated Thoracic Surgery". The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/vats.2017.08.15). The series “Non-intubated Thoracic Surgery” was commissioned by the editorial office without any funding or sponsorship. TCM served as the unpaid Guest Editor of the series. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Goldstein RS, Todd TR, Guyatt G, et al. Influence of lung volume reduction surgery (LVRS) on health related quality of life in patients with chronic obstructive pulmonary disease. Thorax 2003;58:405-10. [Crossref] [PubMed]
- Yusen RD, Lefrak SS, Gierada DS, et al. A prospective evaluation of lung volume reduction surgery in 200 consecutive patients. Chest 2003;123:1026-37. [Crossref] [PubMed]
- Mineo TC, Ambrogi V, Pompeo E, et al. Body weight and nutritional changes after reduction pneumoplasty for severe emphysema: a randomized study. J Thorac Cardiovasc Surg 2002;124:660-7. [Crossref] [PubMed]
- Mineo TC, Ambrogi V, Pompeo E, et al. Impact of lung volume reduction surgery versus rehabilitation on quality of life. Eur Respir J 2004;23:275-80. [Crossref] [PubMed]
- Pompeo E, Marino M, Nofroni I, et al. Reduction pneumoplasty versus respiratory rehabilitation in severe emphysema: a randomized study. Pulmonary Emphysema Research Group. Ann Thorac Surg 2000;70:948-53; discussion 954. [Crossref] [PubMed]
- Mineo D, Ambrogi V, Lauriola V, et al. Recovery of body composition improves long-term outcomes after lung volume reduction surgery for emphysema. Eur Respir J 2010;36:408-16. [Crossref] [PubMed]
- Mineo TC, Pompeo E, Mineo D, et al. Awake nonresectional lung volume reduction surgery. Ann Surg 2006;243:131-6. [Crossref] [PubMed]
- Mineo D, Ambrogi V, Cufari ME, et al. Variations of inflammatory mediators and alpha1-antitrypsin levels after lung volume reduction surgery for emphysema. Am J Respir Crit Care Med 2010;181:806-14. [Crossref] [PubMed]
- Mineo TC, Tamburrini A, Perroni G, et al. 1000 cases of tubeless video-assisted thoracic surgery at the Rome Tor Vergata University. Future Oncol 2016;12:13-8. [Crossref] [PubMed]
- Mineo TC. Epidural anesthesia in awake thoracic surgery. Eur J Cardiothorac Surg 2007;32:13-9. [Crossref] [PubMed]
- Launois C, Barbe C, Bertin E, et al. The modified Medical Research Council scale for the assessment of dyspnea in daily living in obesity: a pilot study. BMC Pulm Med 2012;12:61. [Crossref] [PubMed]
- Ware JE, Snow KK, Kosinski M, et al. SF-36 health survey: manual and interpretation guide. Lincoln, RI: Quality Metric IncoLVRorated, 1993.
- Jones PW, Quirk FH, Baveystock CM, et al. A self-complete measure of health status for chronic airflow limitation. The St. George's Respiratory Questionnaire. Am Rev Respir Dis 1992;145:1321-7. [Crossref] [PubMed]
- Folstein MF, Folstein SE, McHugh PR. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189-98. [Crossref] [PubMed]
- Perbet S, Fernandez-Canal C, Pereira B, et al. Evaluation of Richmond Agitation Sedation Scale According To Alveolar Concentration of Sevoflurane During a Sedation With Sevoflurane in Icu Patients. Intensive Care Med Exp 2015;3:A27. [Crossref]
- Bland JM, Altman DG. Survival probabilities (the Kaplan-Meier method). BMJ 1998;317:1572. [Crossref] [PubMed]
- van Agteren JE, Carson KV, Tiong LU, et al. Lung volume reduction surgery for diffuse emphysema. Cochrane Database Syst Rev 2016;10:CD001001 [PubMed]
- Shah PL, Herth FJ, van Geffen WH, et al. Lung volume reduction for emphysema. Lancet Respir Med 2017;5:147-56. [Crossref] [PubMed]
- Mineo D, Mineo TC. Lung volume reduction surgery: a better understanding for a renewed challenge in the treatment of emphysema. Am J Respir Crit Care Med 2012;186:1068-70. [Crossref] [PubMed]
- Mineo TC, Ambrogi V. Efficacy of awake thoracic surgery. J Thorac Cardiovasc Surg 2012;143:249-50; author reply 250-1. [Crossref] [PubMed]
- DeCamp MM Jr, McKenna RJ Jr, Deschamps CC, et al. Lung volume reduction surgery: technique, operative mortality, and morbidity. Proc Am Thorac Soc 2008;5:442-6. [Crossref] [PubMed]
- Feinleib M, Rosenberg HM, Collins JG, et al. Trends in COPD morbidity and mortality in the United States. Am Rev Respir Dis 1989;140:S9-18. [Crossref] [PubMed]
Cite this article as: Mineo TC, Fabbi E, Ambrogi V. Nonintubated uniportal nonresectional videothoracoscopic lung volume reduction surgery. Video-assist Thorac Surg 2017;2:68.


