Video-assisted anatomical resection for pulmonary blastoma
Introduction
Pulmonary blastomas (PBs) were first described by Barnard in 1952. They represent a rare group of primary lung malignancies accounting for approximately 0.25 to 0.50% of all lung neoplasms (1) and are composed of immature malignant epithelial and/or mesenchymal cells with features resembling embryonic lung tissue.
Surgery is the mainstay of treatment. To date there have been approximately one hundred case reports describing PBs (2). We present two cases of PB resected via a minimally invasive approach.
Case presentation 1
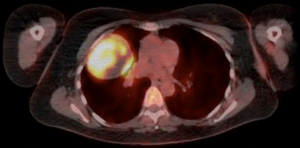
Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images. A 56-year-old Caucasian female with a 26-year history of passive smoking, presented with a six-week history of dry cough and daily haemoptysis. Computed tomography (CT) and positron emission tomography-computed tomography (PET-CT) scans (Figure 1) revealed a well-defined 7.1cm lesion within the upper and middle lobes of the right lung, with suspected involvement of the anterior chest wall and hilar lymphadenopathy. The radiological stage was T3N1M0. Pre-operative lung function tests were within normal limits; FEV1 2.19L (82% predicted), FVC 2.96L (96% predicted) and TLCO 87% of predicted. CT guided biopsy of the pulmonary mass reported hyperchromatic cells and focal small ill-defined acinar structures.
Given the clinical and radiological features of malignancy, a multidisciplinary decision for surgical resection was made.
Procedure
An anterior VATS approach was chosen. The pleural cavity was accessed via two 1 cm incisions and a 4 cm utility incision with soft tissue retraction. A 300 mm × 10 mm Storz Scope® (Karl Storz-endoscope, Germany) was inserted through the camera port and revealed a large tumour occupying the upper and middle lobes with minimal adhesions to the anterior chest wall. The parietal pleura overlying the tumour was not macroscopically infiltrated, therefore an extrapleural dissection of the mass from the chest wall was performed and a frozen section sent which confirmed clear resection margins. A right upper bilobectomy was then performed with systematic hilar and mediastinal lymph nodal dissection (lymph node stations 2R, 4R, 7, 8, 9, 10R, 11R). The specimen was withdrawn intact using Endobag® retrieval system after enlargement of the utility port to 7cm and brief retraction of the ribs. A single chest drain was inserted and left in situ.
Pathology
The tumour was a well circumscribed, haemorrhagic and centrally necrotic mass measuring 8 cm × 7 cm × 8.5 cm, involving the inferior part of the right upper lobe and extending across the fissure into the middle lobe.
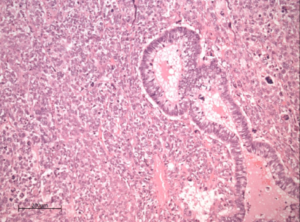
There was no invasion of the pleural surface. A biphasic architecture was demonstrated, formed by a glandular tubular structure mixed with a more prominent stromal component. These appearances were consistent with a diagnosis of biphasic PB (Figure 2). The pathological stage was pT3 (PL1) N0.
Follow-up
No immediate complications were noted and the patient was discharged home on post-operative day two. The patient was offered adjuvant chemotherapy however declined. To date routine CT scans have revealed no evidence of recurrence 3 years from surgery.
Case presentation 2
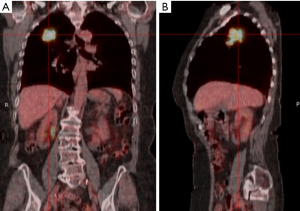
Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images. A 78-year-old woman presented with cough, haemoptysis and weight loss. She had a background of severe emphysema, type 2 diabetes mellitus and osteoarthritis. Chest radiograph revealed an opacity in the right upper zone and CT confirmed a 4.5 cm lesion in the right upper lobe with no evidence of mediastinal lymph node involvement. CT-PET confirmed no evidence of regional or distant spread, therefore clinically staged ascT2bN0M0 (Figure 3). A CT-guided biopsy provided a provisional diagnosis of carcinosarcoma. Performance status was 1 and pulmonary function tests were within normal range, therefore a multi-disciplinary team concluded that surgical resection was indicated.
Procedure
An uncomplicated three-port VATS right upper lobectomy and lymphadenectomy was performed via an anterior approach as described above.
Pathology
The tumour was partially necrotic, consisting of malignant glands set within a malignant cell stroma. In other areas the tumour showed sheets of undifferentiated blastoma-like areas, in keeping with a diagnosis of pulmonary blastoma. The tumour did not involve the visceral pleura and resection margins were clear. Mediastinal lymph nodes were uninvolved. Pathological stage was pT2bN0. Uninvolved lung parenchyma showed significant emphysematous changes.
Follow-up
Post-operative recovery was complicated by prolonged air-leak and surgical emphysema. The patient was discharged 20 days following surgery with an ambulatory chest drain in-situ, which was removed the following week at outpatient clinic. A multidisciplinary meeting decided that patient is not for adjuvant chemotherapy due to stage, complete resection, age and co-morbidities. Regular follow up with CT has revealed no evidence of recurrence to date, 3 years on from surgery.
Discussion
The current WHO classification describes five sub-groups of sarcomatoid carcinoma of the lung including pulmonary blastoma, pleomorphic carcinoma, spindle cell carcinoma, giant cell carcinoma and carcinosarcoma. These tumours are poorly differentiated non-small cell lung cancer, which contain a component of sarcoma/sarcoma-like differentiation.
PB is a biphasic tumour containing a primitive epithelial component and a primitive mesenchymal stroma. Preoperative diagnosis on the basis of cytology or small biopsies alone is often inaccurate due to histological heterogeneity and accurate diagnosis is often made following surgical excision. Differentiation of PB from foetal adenocarcinoma, pleuropulmonary blastoma or primary/secondary sarcomas is challenging, immunohistochemical and molecular studies in addition to morphological features aid to differentiate theses tumours (3). There is no general consensus regarding their optimal management and prognosis, however, surgical resection is the mainstay of treatment (2). The use of minimally invasive techniques for the resection of PBs has not been described in the literature to date; the two cases above illustrate the successful use of VATS for the complete resection of PB. PBs tend to present with non-specific respiratory symptoms that can mimic a lower respiratory tract infection.
Approximately 40% of cases are asymptomatic and are incidentally found on chest X-ray (4). In these cases, the clinical onset was characterised by persistent symptoms of cough, dyspnoea, haemoptysis and weight loss. It has been demonstrated that PBs have a female preponderance and are associated with cigarette smoking in 82% of cases (2). They are predominantly tumours of adulthood with the peak incidence in the fourth decade.
When compared to other primary lung tumours, PBs tend to be more aggressive and are commonly located in the peripheries of the lungs, only in 25% of cases is endobronchial growth demonstrated. Median survival rates described are 25% at 1 year and 16% at 5 years (5). Several factors indicate a poor prognosis, including biphasic histology, metastases at initial presentation, tumours greater than five centimetres and tumour recurrence (2).
A study conducted by Liman et al. found that PBs are infrequently diagnosed pre-operatively despite the use of percutaneous and transbronchial biopsies or more invasive procedures such as mediastinoscopy and VATS due to the extensive necrosis of the lesions and lack of cellular material (6). This finding was confirmed in our cases.
The literature addressing the use and efficacy of adjuvant chemotherapy and radiotherapy is limited, likely due to the infrequency of this malignancy. Consequently, chemotherapy regimens appear to be individualized based on the histopathology and stage of tumour, the literature suggests that a platinum based regimen is the most frequent choice (1).
The use of VATS for pulmonary lobectomies is well established and is considered to be minimally invasive compared to open thoracotomy, therefore reducing the impact of surgery on patients. To date, there is no evidence in the literature regarding the use of VATS for the excision of PB. It is likely that this is due to the rarity of the disease and frequent advanced stage at presentation. The two cases reported here were carried out at separate institutions. In both cases the resection of the tumour was performed via a minimal access approach in order to provide the patients with the associated benefits, without compromising oncological principles.
PBs are a rare type of advanced primary lung tumour with surgical excision as the first line of treatment. Adjuvant chemotherapy is described due to the high risk of recurrence and aggressive behaviour of the tumour; however, there is little data to evaluate its efficacy based upon the rarity of the disease. A co-ordinated multidisciplinary approach from surgeons and oncologists can achieve promising mid-term results. The two cases presented illustrate the safe and effective use of VATS for complete excision of PB.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/vats.2017.02.06). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Larsen H, Sørensen JB. Pulmonary blastoma: a review with special emphasis on prognosis and treatment. Cancer Treat Rev 1996;22:145-60. [Crossref] [PubMed]
- Alahwal MS, Maniyar IH, Saleem F, et al. Pulmonary blastoma: a rare primary lung malignancy. Case Rep Med 2012;2012:471613.
- Travis WD, Brambilla E, Nicholson AG, et al. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J Thorac Oncol 2015;10:1243-60. [Crossref] [PubMed]
- Walker RI, Suvarna K, Matthews S. Case report: pulmonary blastoma: presentation of two atypical cases and review of the literature. Br J Radiol 2005;78:437-40. [Crossref] [PubMed]
- Robert J, Pache JC, Seium Y, et al. Pulmonary blastoma: report of five cases and identification of clinical features suggestive of the disease. Eur J Cardiothorac Surg 2002;22:708-11. [Crossref] [PubMed]
- Liman ST, Altinok T, Topcu S, et al. Survival of biphasic pulmonary blastoma. Respir Med 2006;100:1174-9. [Crossref] [PubMed]
Cite this article as: Abah U, Mahendran H, Scarci M, Rassl D, Bartosik W, Granato F. Video-assisted anatomical resection for pulmonary blastoma. Video-assist Thorac Surg 2017;2:13.