
Subxiphoid VATS approach for the mediastinum
Introduction
Most of general thoracic surgical operations are performed through the traditional thoracotomy or sternotomy or with modern video-assisted thoracic surgery (VATS) approach. Significant severity of pain is a common disadvantage of all these procedures, even in case of VATS. There are several sources of the pain after thoracotomy and VATS but irritation of injury of the intercostal nerves is probably the most important one. To overcome the problem of severe postoperative pain a subxiphoid approach for various thoracic surgical procedures has been developed in the last two decades.
Initially, in the 1970s the subxiphoid approach was introduced in diagnosis and treatment of the pericardial fluids including hematoma in suspicion of the heart trauma and pericarditis (1). Afterwards, the subxiphoid approach was used for the other intrathoracic procedures gaining a special importance for use of thymectomy and more recently for minimally invasive uniportal VATS pulmonary resections (2-4).
The other uses of the subxiphoid approach included cardiac procedures (mainly for cardiac tamponade, pericardial fluid, but also for mitral valve surgery, pericardial atrial ablation, coronary by-pass surgery and dissection of the internal thoracic artery), the Nuss procedures, thoracic sympathectomy, drainage of the mediastinum in the necrotizing mediastinitis, laparoscopic diaphragmatic hernia repair and esophageal resection (5-13).
We performed a search on PUBMED with keywords: subxiphoid thoracic surgery, subxiphoid mediastinal surgery, infrasternal mediastinal surgery and found 51 articles on the use of the subxiphoid (infrasternal) approach for all types of operations, including 24 articles on thymectomy for myasthenia gravis (MG) and thymomas, resection of the other mediastinal tumors and the mediastinal metastases, mediastinal lymph nodes and parathyroid glands.
The subxiphoid approach can be solely as a uniportal approach, or can be combined with the other minimally invasive incisions like intercostal or subcostal VATS ports, or the transcervical incision (3,14,15). In case of esophageal resection the subxiphoid approach can be combined with laparoscopy (11).
Types of the surgical procedures of the mediastinum performed through the subxiphoid approach are listed in Table 1.
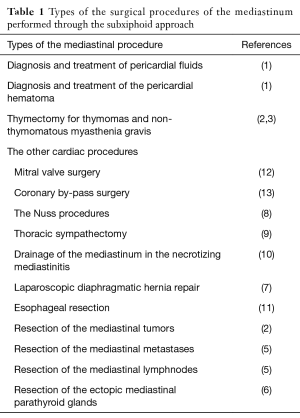
Full table
There are several variants of the subxiphoid approach for thymectomy. It can be used as a sole approach as was originally described by Kido et al., or it can be combined with additional intercostal or subcostal VATS porta and transcervical incision (2,3,15). Thymectomy can be performed inside the mediastinum, or with opening of one or both pleural cavities to improve the view and increase completeness of dissection. Suda et al. were able to perform robotic thymectomy through the subxiphoid/bilaleral VATS approach (14). The subxiphoid approach for thymectomy has several advantages in comparison to the wide-spreaded unilateral VATS approach. Besides of being less painful it provides a bilateral view of the mediastinum enabling clear visualization of both phrenic nerves, which is a prerequisite of complete extended thymectomy. The useful technical maneuver described by our team and afterwards by Takeo et al. is double elevation of the sternum, which greatly improves the view of the mediastinum (3,16). The other technique to improve the view of the mediastinum during subxiphoid thymectomy is to use a Single Incision Laparoscopy plate (SILS) with carbon dioxide (CO2) insufflation (14). Our previously reported institutional results of 5-year complete remission rates of MG after thymectomy showed that the transcervical-subxiphoid bilateral VATS thymectomy provided comparable results to the extended transsternal thymectomy (17).
The recently results of the randomized trial of thymectomy in MG supported a beneficial use of thymectomy for non-thymomatous MG, but only with the use of the extended transsternal approach similar to this described by Jaretzki et al. (18,19). The need for a randomized trial comparing the effectiveness of extended transsternal and minimally invasive techniques of thymectomy is apparent.
The use of subxiphoid approach for the awake endoscopic thymectomy with a sternal lifting was also described (20).
In case of thymectomy, the subxiphoid approach can be combined with a transcervical incision and bilateral single-port VATS to achieve a maximal extensiveness of dissection (we call the procedure “transcervical-subxiphoid VATS maximal thymectomy”) (3).
The aim of this article is to present our current technique utilizing the subxiphoid approach in surgery of the mediastinum, specifically in thymectomy for non thymomatous myasthenia gravis, early stage thymomas and repeated thymectomy (re-thymectomy) for recurrent myasthenia.
Patient selection and workup
Patients with non thymomatous MG, The Masaoka stage I-III thymomas and rethymectomies for previous thymectomy are suitable candidates for an operation performed through the subxiphoid approach. Written informed consent was obtained from all patients.
Standard blood and spirometric tests for patients undergoing thoracic operation are mandatory. The other requirements for surgery is a reliable confirmation of diagnosis of MG and a CT scan of the chest.
Pre-operative preparation
In case of MG a proper stabilization of the disease with anticholinesterase, steroids/immunosuppressive drugs and/or plasmapheresis/intravenous immunoglobulins is crucial to avoid postoperative myasthenic crisis.
Equipment preference card
Special surgical instruments necessary for subxiphoid VATS extended thymectomy:
- The Rochard frame with two one-tooth hooks for elevation of the sternum from the sides of the manubrium and the lower angle of the sternum;
- The Yankauer suction tube (or similar stiff metal suction tube);
- The bariatric extra-long laparoscopic ports (in case of the subxiphoid-subcostal approach);
- The bipolar coagulation or harmonic knife or ligasure device.
Procedure
Surgical technique of an extended transcervical approach utilizes a typical 5-8 centimeters subxiphoid below the lower angle of the sternum. The medial parts of both abdominal rectus muscles are cut transversely with electrocautery to expose the xiphoid process and the costal arches bilaterally. The xiphoid process is removed in case of ossification, but in the case of young patients when the process is still soft and cartilaginous it can be simply disconnected from the bone of the lower sternal angle without removal. The next maneuver facilitating access to the chest is elevation of the lower angle of the sternum with a hook connected to the modified Rochard frame (Asculap-Chifa company). Elevation of the lower angle of the sternum creates the space between the chest wall and the diaphragm and the pericardium to enter the chest cavity after division of the mediastinal pleura. Usually, the right pleural cavity is entered first after ventilation of the right lung is disconnected.
There are at least three alternative routes to introduce a thoracoscopy to the chest cavity. One is the subxiphoid incision, the others are an intercostal incision and a subcostal incisions. The last option deserves description. The possible advantage of the subcostal incision is less pain in comparison to the intercostal approach. It is an imperative to avoid entrance to the abdominal cavity. To achieve this aim we use an original technique. We perform a 1–1.5 cm incision a little below and parallel to the costal margin in medially to the mid-clavicular line. We introduce the Yankauer suction tube and the Cameleon thoracoscopy (Storz, Germany) through the subxiphoid incision. The Yankauer suction tube presses the dome of the diaphragm in the caudal direction, which stretches the diaphragm and allows to avoid violation of the diaphragmatic peritoneum. Then to introduce a dissector to enter the pleural space just below the insertion of the diaphragm to the chest wall. This maneuver is done under control of the Cameleon thoracoscopy. We use this specific type of thoracoscopy because of the extremely wide range of view. After the channel to the chest wall is made with a dissector this instrument is withdrawn and the extra-long bariatric port is introduced to the chest cavity, again under control of the Cameleon thoracoscopy (Figure 1). In the same way a bariatric port is introduced to the other pleural cavity, if necessary. The Cameleon thoracoscopy is inserted to the right pleural cavity for control of dissection of the thymus and the adipose tissue of the mediastinum, which is removed en bloc with the whole thymus and thymoma (if present). The instruments used for dissection are a Yankauer suction tube, cautery device (electrocautery, bipolar cautery, harmonic knife or Ligasure). These instruments and a ring forceps, if necessary are introduced through the subxiphoid incision. The specimen is dissected from the inner surface of the sternum. At this stage the second, one tooth hook is inserted under the sternal manubrium and connected to the Rochard frame to elevate the sternum from the upper part (Figure 2). Elevation of the sternum, both from the manubrium and the lower angle improves the view of the chest and facilitates dissection and visualization of the structures of the mediastinum, especially in the area of the lower poles of the thyroid, which are otherwise inaccessible from the chest. Our current policy is to start dissection from the right pleural cavity and to open the left mediastinal pleura on the relatively stage of the operation. This maneuver allows for dissection on both sides of the anterior mediastinum simultaneously, which is especially helpful in case of the obese patients with a large volume of the mediastinal fat. In such cases it is very helpful to transfer the whole dissected specimen to the opposite pleural cavity which clears the view of the mediastinum. Generally, simultaneous opening of both pleural cavities necessitates ventilation of both lungs, but in practice it is possible to proceed with the ventilation of the left lung while the ventilation of the right lung is disconnected. The specimen containing the thymus is dissected from the caudal towards the cranial direction and both phrenic nerves are the lateral margins of dissection. The left innominate vein is visualized and the thymic and the inferior thyroid veins are dissected, clipped and divided. The left innominate vein and the innominate artery are dissected and followed cranially. The pretracheal fat is removed with visualization of the anterior wall of the trachea (Figure 3). The right and left upper poles of the thymus are dissected from the lateral sides. Dissection is proceeded cranially until the lower poles of the thyroid glands are visualized (Figure 4). At this level both thyro-thymic ligaments are divided and the specimen is removed in the endobag (Figure 5). Removal of the fatty tissue from the aorta-pulmonary window is the last step of the operation. One chest tube is inserted to each pleural cavity through the ports which are removed afterwards. The hooks elevating the sternum are removed and the subxiphoid incision is closed in the standard manner.





Role of team members
To manage effectively patients undergoing thymectomy for MG there must be a well organized team consisting of several specialists experienced in diagnosis, operative treatment and postoperative care of patients with MG:
- Neurologists—diagnosis of MG, preoperative preparation of the MG patient with anticholinesterase, steroid/immunosuppressive drugs or/and plasmapheresis/intravenous immunoglobulins, selection of patients for thymectomy (together with a thoracic surgeon), follow-up, further care of MG patients;
- Thoracic surgeons—selection of patients for thymectomy (together with a neurologist), performance of an operation in minimally invasive technique;
- Anaesthesiologists—proper conducting of anaesthesia and postoperative treatment of myasthenic patient.
- Nurses—care of MG patients before and after an operation.
- Physiotherapists—care of MG patients before and after an operation.
Tips, tricks and pitfalls
There are several tips and tricks to perform a subxiphoid-VATS extended thymectomy successfully:
- A patients is positioned supine instead of a decubitus or semilateral position used for unilateral VATS thymectomy. A supine position enables approach for both sides of the mediastinum with clear visualization of both phrenic nerves, which is a prerequisite of a complete and extensive removal of the fatty tissue of the mediastinum.
- Elevation of the sternum enables visualization of the structures of the superior mediastinum and the lower neck, which makes it possible to proceed dissection up to the level of the lower poles of the thyroid gland. Without division of the thyro-thymic ligaments at the level of the lower poles of the thyroid completeness of thymectomy is questionable.
- The use of bipolar electrocautery, harmonic knife or ligasure.
- The use of the Yankauer curved stiff metallic suction tube is extremely convenient both for suction and a dissection of the mediastinal suction. Additionally, in case of moderate bleeding pressure of the tip of the suction tube can control bleeding and facilitate application of a vascular clip, or management of the bleeding with bipolar cautery (or similar devices).
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Luca Bertolaccini and Piergiorgio Solli) for the series “VATS: the age of maturity” published in Video-Assisted Thoracic Surgery. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/vats.2016.12.02). The series “VATS: the age of maturity” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from all patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Santos GH, Frater RW. The subxiphoid approach in the treatment of pericardial effusion. Ann Thorac Surg 1977;23:467-70. [Crossref] [PubMed]
- Kido T, Hazama K, Inoue Y, et al. Resection of anterior mediastinal masses through an infrasternal approach. Ann Thorac Surg 1999;67:263-5. [Crossref] [PubMed]
- Zieliński M. Technique of transcervical--subxiphoid-vats "maximal" thymectomy in treatment of myasthenia gravis. Przegl Lek 2000;57:64-5. [PubMed]
- Song N, Zhao DP, Jiang L, et al. Subxiphoid uniportal video-assisted thoracoscopic surgery (VATS) for lobectomy: a report of 105 cases. J Thorac Dis 2016;8:S251-7. [PubMed]
- Arom KV, Franz JL, Grover FL, et al. Subxiphoid anterior mediastinal exploration. Ann Thorac Surg 1977;24:289-90. [Crossref] [PubMed]
- Wei JP, Gadacz TR, Weisner LF, et al. The subxiphoid laparoscopic approach for resection of mediastinal parathyroid adenoma after successful localization with TC-99m-sestamibi radionuclide scan. Surg Laparosc Endosc 1995;5:402-6. [PubMed]
- Borrego Galán JC, Alcázar Montero MC, García García J, et al. Morgagni's diaphragmatic hernia. Report of 2 cases. Rev Esp Enferm Apar Dig 1989;75:389-92. [PubMed]
- Becmeur F, Ferreira CG, Haecker FM, et al. Pectus excavatum repair according to Nuss: is it safe to place a retrosternal bar by a transpleural approach, under thoracoscopic vision? J Laparoendosc Adv Surg Tech A 2011;21:757-61. [Crossref] [PubMed]
- Chen JT, Liao CP, Chiang HC, et al. Subxiphoid single-incision thoracoscopic bilateral ablative sympathectomy for hyperhidrosis. Interact Cardiovasc Thorac Surg 2015;21:119-20. [Crossref] [PubMed]
- Guan X, Zhang WJ, Liang X, et al. Optimal surgical options for descending necrotizing mediastinitis of the anterior mediastinum. Cell Biochem Biophys 2014;70:109-14. [Crossref] [PubMed]
- Xu M, Liu C, Guo M, et al. Combined laparoscopic-thoracoscopic esophagectomy and intrathoracic esophagogastric anastomsis. J Thorac Dis 2013;5:895-7. [PubMed]
- Watanabe G, Yamaguchi S, Tomiya S, et al. Awake subxyphoid minimally invasive direct coronary artery bypass grafting yielded minimum invasive cardiac surgery for high risk patients. Interact Cardiovasc Thorac Surg 2008;7:910-2. [Crossref] [PubMed]
- Karagoz HY, Bayazit K, Battaloglu B, et al. Minimally invasive mitral valve surgery: the subxiphoid approach. Ann Thorac Surg 1999;67:1328-32; discussion 1333. [Crossref] [PubMed]
- Suda T, Kaneda S, Hachimaru A, et al. Thymectomy via a subxiphoid approach: single-port and robot-assisted. J Thorac Dis 2016;8:S265-71. [PubMed]
- Zhao J, Wang J, Zhao Z, et al. Subxiphoid and subcostal arch thoracoscopic extended thymectomy: a safe and feasible minimally invasive procedure for selective stage III thymomas. J Thorac Dis 2016;8:S258-64. [PubMed]
- Takeo S, Sakada T, Yano T. Video-assisted extended thymectomy in patients with thymoma by lifting the sternum. Ann Thorac Surg 2001;71:1721-3. [Crossref] [PubMed]
- Zielinski M, Hauer L, Hauer J, et al. Comparison of complete remission rates after 5 year follow-up of three different techniques of thymectomy for myasthenia gravis. Eur J Cardiothorac Surg 2010;37:1137-43. [Crossref] [PubMed]
- Wolfe GI, Kaminski HJ, Aban IB, et al. Randomized Trial of Thymectomy in Myasthenia Gravis. N Engl J Med 2016;375:511-22. [Crossref] [PubMed]
- Jaretzki A 3rd, Wolff M. "Maximal" thymectomy for myasthenia gravis. Surgical anatomy and operative technique. J Thorac Cardiovasc Surg 1988;96:711-6. [PubMed]
- Matsumoto I, Oda M, Watanabe G. Awake endoscopic thymectomy via an infrasternal approach using sternal lifting. Thorac Cardiovasc Surg 2008;56:311-3. [Crossref] [PubMed]
- Zieliński M, Rybak M, Solarczyk-Bombik K, et al. Subxiphoid incision, opening of the right pleural cavity and introduction of the bariatric port through the subcostal space. Asvide 2017;4:001. Available online:
- Zieliński M, Rybak M, Solarczyk-Bombik K, et al. Introduction of the hook elevating the manubrium. Asvide 2017;4:002. Available online:
- Zieliński M, Rybak M, Solarczyk-Bombik K, et al. Dissection of the left lower pole of the thyroid gland, inferior thyroid vein, pretracheal fat and the thymic vein. Asvide 2017;4:003. Available online:
- Zieliński M, Rybak M, Solarczyk-Bombik K, et al. The view of the upper mediastinum and the lower neck after dissection of the upper poles of the thymus from the thyroid gland. Asvide 2017;4:004. Available online:
- Zieliński M, Rybak M, Solarczyk-Bombik K, et al. Dissection of the fatty tissue from the pericardium and the left phrenic nerve and removal of the specimen in the endobag. Asvide 2017;4:005. Available online:
Cite this article as: Zieliński M, Rybak M, Solarczyk-Bombik K, Wilkojc M, Czajkowski W, Kosinski S, Fryzlewicz E, Nabialek T, Pankowski J. Subxiphoid VATS approach for the mediastinum. Video-assist Thorac Surg 2017;2:1.


